|
|
|
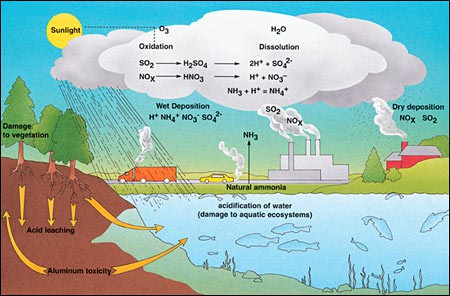
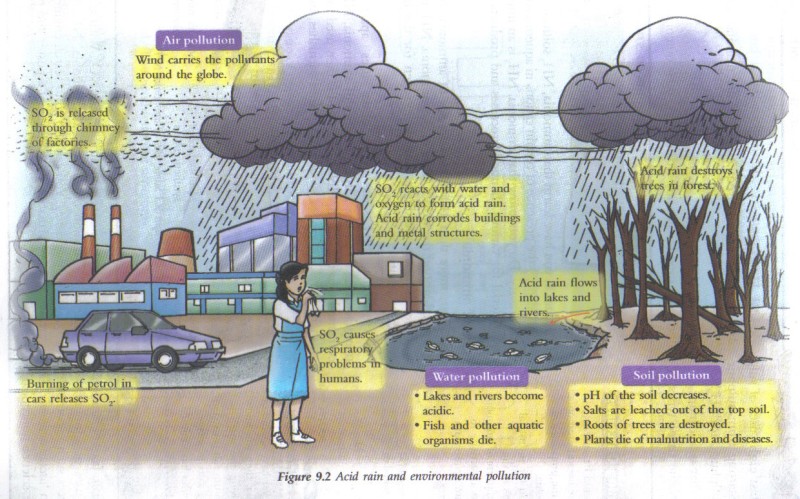
Acid Deposition"Acid rain" is a broad term referring to a mixture of wet and dry deposition (deposited material) from the atmosphere containing higher than normal amounts of nitric and sulfuric acids. The precursors, or chemical forerunners, of acid rain formation result from both natural sources, such as volcanoes and decaying vegetation, and man-made sources, primarily emissions of sulfur dioxide (SO2) and nitrogen oxides (NOx) resulting from fossil fuel combustion. In the United States, roughly 2/3 of all SO2 and 1/4 of all NOx come from electric power generation that relies on burning fossil fuels, like coal. Acid rain occurs when these gases react in the atmosphere with water, oxygen, and other chemicals to form various acidic compounds. The result is a mild solution of sulfuric acid and nitric acid. When sulfur dioxide and nitrogen oxides are released from power plants and other sources, prevailing winds blow these compounds across state and national borders, sometimes over hundreds of miles.
source: http://www.epa.gov/acidrain/what/index.html |
How much SO2 and NOx is being pumped into the atmosphere?
54% of Hong Kong's energy comes from COAL. * Timelapse Coal Power Plant from Jeff Grewe on Vimeo. |
|

Reading: Davis and Nagle:
pg253-258 + Questions 1-5 | ||||||||
Images Source: http://www.eoearth.org/files/185201_185300/185231/sfc-water-quality.jpg
The effects of acid deposition on soil, water and living organisms (5.8.2)
Acid depositions can have direct effects e.g. by weakening tree growth, toxic effects e.g. effect of aluminum ions on fish and plant roots and nutrient effects e.g. leaching of nutrients.
Describe these three examples:
Effect of acid rain on coniferous forests
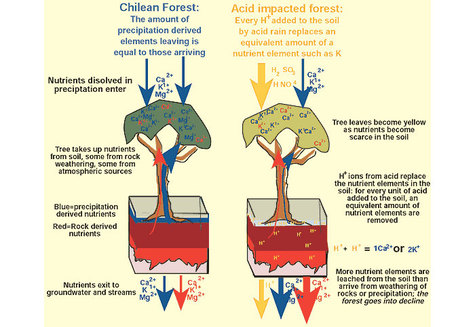
Acid rain damage became a focus of environmental attention in the early 1970s when Germany’s Black Forest showed a dieback when trees of all ages, both coniferous and deciduous showed signs of physical damage. Acid rain affects forest biology in several ways. Leaves and buds show yellowing (loss of chlorophyll) and damage in the form of lesions, thinning of wax cuticles. These and other changes reduce growth, allow nutrients to be leached out and washed away and pathogens and insects to gain entry. Symbiotic root microbes are killed and this greatly reduces the availability of nutrients further reducing tree growth. Acid rain affects the soil by reducing the ability of soil particles to hold on to nutrients, such as calcium, magnesium and potassium ions which are then leached out. Furthermore the acid releases toxic alumnum ions from soil particles which then damage root hairs. Trees can thus be weakened to the point of death.
Toxic effect of aluminum ions on fish and other aquatic organisms; lichens
Rain washes aluminium ions released from soil into streams and lakes and leads to the death of aquatic organisms. Fish are particularly sensitive. Aluminium reacts with mucus on the gills of fish. This inhibits the normal intake of oxygen and salt. Fish gasp for breath and the salt content of their bodies is slowly lost, leading to death.
Lichens which are primative plants found growing on trees and buldings are particularly sensitive to gaseous pollutants like sulphur dixide and are used as indirect measures of pollution. Immediately downwind of a heavily polluting industrial region only a few tolerant species are found. These are indicator species of high levels of air pollution. As the distance from the source of pollutants increases more and more species are able to survive. Tables of lichen indicator species are used to estimate pollution levels.
Nutrient removal effect on soil fertility
As described above acid rain affects the soil by reducing the ability of soil particles to hold on to nutrients, such as calcium, magnesium and potassium ions which are then leached out. Acid rain also inhibits nitrogen-fixing bacteria and this their ability to add
Lake acidification
Begins with the deposition of the byproducts acid precipitation (SO4 and H ions) in terrestrial areas located adjacent to the water body. Hydrologic processes then move these chemicals through soil and bedrock where they can react with limestone and aluminum-containing silicate minerals. After these chemical reactions, the leachate continues to travel until it reaches the lake. The acidity of the leachate entering lake is controlled by the chemical composition of the effected lake's surrounding soil and bedrock. If the soil and bedrock is rich in limestone the acidity of the infiltrate can be reduced by the buffering action of calcium and magnesium compounds. Toxic aluminum (and some other toxic heavy metals) can leach into the lake if the soil and bedrock is rich in aluminum-rich silicate minerals. (Source:PhysicalGeography.net)
Acid depositions can have direct effects e.g. by weakening tree growth, toxic effects e.g. effect of aluminum ions on fish and plant roots and nutrient effects e.g. leaching of nutrients.
Describe these three examples:
Effect of acid rain on coniferous forests
Acid rain damage became a focus of environmental attention in the early 1970s when Germany’s Black Forest showed a dieback when trees of all ages, both coniferous and deciduous showed signs of physical damage. Acid rain affects forest biology in several ways. Leaves and buds show yellowing (loss of chlorophyll) and damage in the form of lesions, thinning of wax cuticles. These and other changes reduce growth, allow nutrients to be leached out and washed away and pathogens and insects to gain entry. Symbiotic root microbes are killed and this greatly reduces the availability of nutrients further reducing tree growth. Acid rain affects the soil by reducing the ability of soil particles to hold on to nutrients, such as calcium, magnesium and potassium ions which are then leached out. Furthermore the acid releases toxic alumnum ions from soil particles which then damage root hairs. Trees can thus be weakened to the point of death.
Toxic effect of aluminum ions on fish and other aquatic organisms; lichens
Rain washes aluminium ions released from soil into streams and lakes and leads to the death of aquatic organisms. Fish are particularly sensitive. Aluminium reacts with mucus on the gills of fish. This inhibits the normal intake of oxygen and salt. Fish gasp for breath and the salt content of their bodies is slowly lost, leading to death.
Lichens which are primative plants found growing on trees and buldings are particularly sensitive to gaseous pollutants like sulphur dixide and are used as indirect measures of pollution. Immediately downwind of a heavily polluting industrial region only a few tolerant species are found. These are indicator species of high levels of air pollution. As the distance from the source of pollutants increases more and more species are able to survive. Tables of lichen indicator species are used to estimate pollution levels.
Nutrient removal effect on soil fertility
As described above acid rain affects the soil by reducing the ability of soil particles to hold on to nutrients, such as calcium, magnesium and potassium ions which are then leached out. Acid rain also inhibits nitrogen-fixing bacteria and this their ability to add
Lake acidification
Begins with the deposition of the byproducts acid precipitation (SO4 and H ions) in terrestrial areas located adjacent to the water body. Hydrologic processes then move these chemicals through soil and bedrock where they can react with limestone and aluminum-containing silicate minerals. After these chemical reactions, the leachate continues to travel until it reaches the lake. The acidity of the leachate entering lake is controlled by the chemical composition of the effected lake's surrounding soil and bedrock. If the soil and bedrock is rich in limestone the acidity of the infiltrate can be reduced by the buffering action of calcium and magnesium compounds. Toxic aluminum (and some other toxic heavy metals) can leach into the lake if the soil and bedrock is rich in aluminum-rich silicate minerals. (Source:PhysicalGeography.net)
Measuring Acid Deposition
* Source: HKSAR Environmental Bureau
Acid rain resources by Darlene Oehlke
|
|
| ||||||||||||||||||